In the aerospace industry, the integrity of critical components is paramount, and among these, aluminum alloy shell castings play a vital role in applications such as aero-engine housings. These shell castings must exhibit exceptional mechanical properties, internal soundness, and surface quality to withstand rigorous operational conditions. However, during manufacturing, a peculiar phenomenon has been observed: linear defects that appear post-machining, often detected through fluorescent inspection. These defects, initially suspected as cracks, have raised concerns regarding the structural reliability of shell castings. In this comprehensive analysis, I delve into the nature, origin, and mitigation strategies for these linear defects, drawing from extensive experimental investigations and metallurgical principles. The focus remains on aluminum alloy shell castings, a term I will reiterate to emphasize the context, as understanding these defects is crucial for advancing casting technologies and ensuring component safety.
The shell castings in question are typically thin-walled with complex geometries, such as those used in engine compartments. For instance, a representative shell casting might feature an outer diameter of approximately 200 mm, a height of 180 mm, and a multi-layered structure with inner and outer walls connected by numerous blades. The wall thickness can be as minimal as 2.6 mm, with blades forming cavities of about 5.6 mm gaps. This intricate design, while optimizing performance, introduces challenges in casting, including high flow resistance during mold filling and susceptibility to defects like inclusions. To visualize such a structure, consider the following representation, which highlights the complexity of these shell castings:
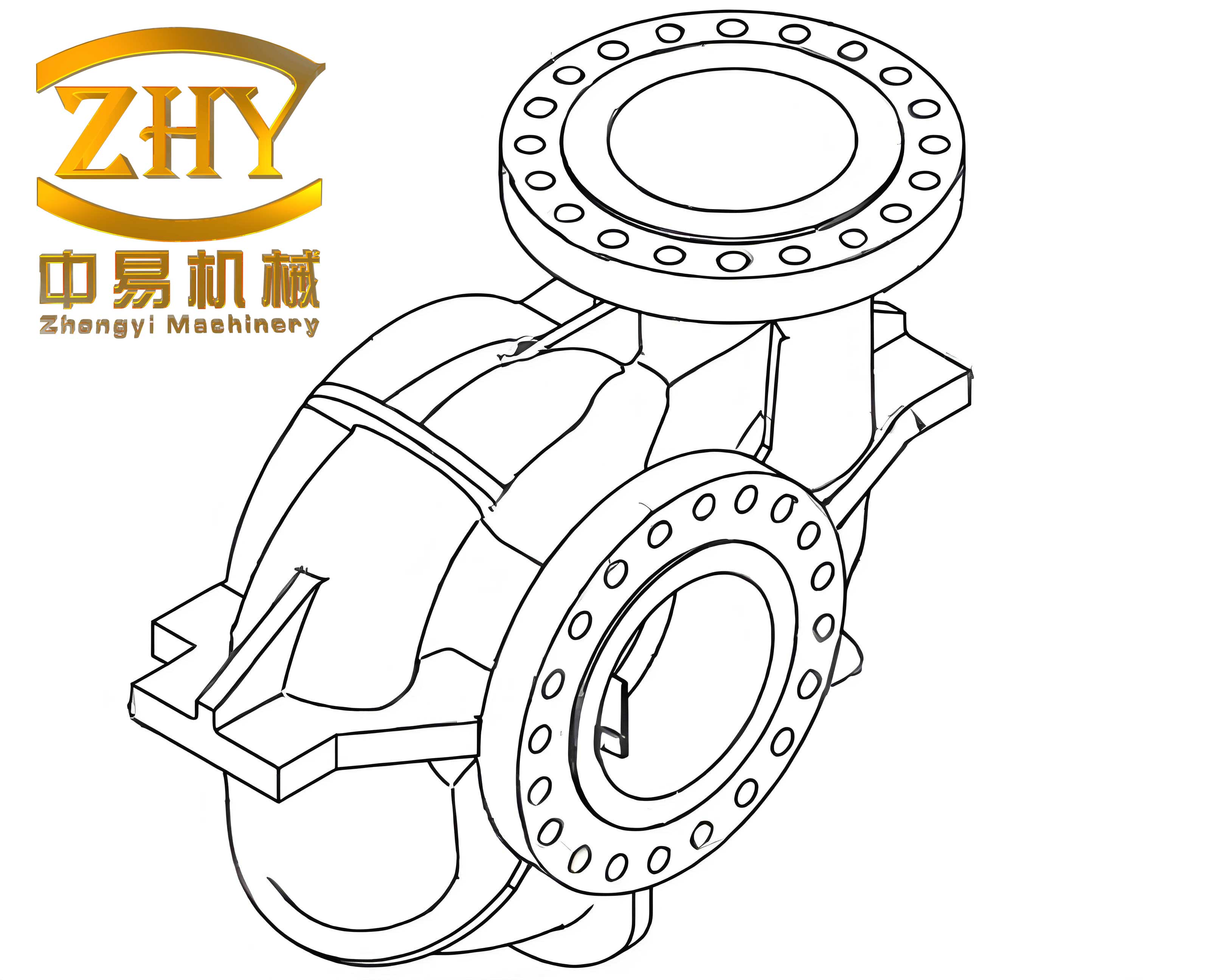
The linear defects often manifest at the roots of blades after machining, appearing as straight or slightly curved lines under macroscopic examination. Initially, these were misinterpreted as cracks due to their resemblance to stress-induced fractures. However, through meticulous analysis, I have determined that these are not true cracks but rather the surface manifestation of internal oxide inclusions subjected to external forces during processing. This distinction is critical, as it shifts the focus from material failure to process-induced imperfections in shell castings. In this article, I will explore the experimental methodologies, present detailed results with tables and formulas, discuss the root causes, and propose preventive measures to enhance the quality of aluminum alloy shell castings.
My investigation began with the selection of shell castings exhibiting typical linear defects, as identified via fluorescent testing. These defects were predominantly located at the blade roots on the pressure side. Macro-observation using stereo microscopy revealed that the defects were relatively straight and aligned with the blade geometry, lacking the irregular patterns often associated with cracking. To probe deeper, I employed scanning electron microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDS) on fractured surfaces. The samples were carefully sectioned and fractured to expose the defect interfaces, which were then cleaned and analyzed. This approach allowed for a multi-scale examination, from macroscopic features to microscopic details, essential for understanding the defect morphology in shell castings.
The SEM analysis of the fractured surfaces revealed three distinct regions, each with unique characteristics. Region 1, adjacent to the linear defect on the blade root surface, exhibited a wrinkled or folded morphology, devoid of typical fracture features like cleavage or ductile dimples. This suggested that the defect was not a crack but rather an interface where material had been displaced. Region 2, located within the blade interior and partially connected to Region 1, showed similar wrinkled surfaces, often with a flower-like pattern in backscattered electron images. At the core of these patterns, particulate inclusions were evident. Region 3, resulting from intentional fracturing, displayed classic ductile dimples, indicative of normal material behavior. The EDS composition analysis of these regions provided further insights, as summarized in the table below:
| Region | O (wt%) | Al (wt%) | Si (wt%) | Cu (wt%) | Mg (wt%) | Remarks |
|---|---|---|---|---|---|---|
| Region 1 | 20.77 | 48.73 | 21.96 | 1.28 | 3.55 | Wrinkled surface, high O and Mg |
| Region 2 | 19.63 | 60.70 | 16.03 | 1.56 | 3.80 | Flower-like pattern, inclusions present |
| Region 3 | 1.52 | 79.58 | 16.88 | 2.09 | – | Ductile dimples, low O |
| Inclusion | 44.18 | 40.01 | 15.21 | – | 2.50 | Particulate oxide, very high O |
The data clearly indicate elevated oxygen and magnesium levels in Regions 1 and 2, along with the inclusions, pointing to the presence of oxide phases. In contrast, Region 3 shows low oxygen content, consistent with the base aluminum alloy. This compositional gradient confirms that the linear defects are associated with oxide inclusions, likely formed during melting or pouring. The wrinkled morphology arises from the deformation of these brittle oxides under mechanical stress during machining, causing them to become visible on the surface of shell castings. Thus, the defect is essentially an embedded oxide film that becomes apparent post-processing, rather than a crack propagating from stress.
To understand the formation of these oxide inclusions, I must delve into the metallurgy of aluminum alloys. Aluminum has a high affinity for oxygen, leading to the rapid formation of alumina (Al2O3) on molten metal surfaces. In ideal conditions, this oxide layer is dense and protective, preventing further oxidation. However, during the melting and casting of shell castings, several factors can disrupt this layer. The process involves adding alloying elements, such as magnesium and silicon, via master alloys or recycled material, necessitating stirring to ensure homogeneity. Improper stirring can entrain the surface oxide into the bulk melt, introducing inclusions. Moreover, other elements like magnesium can form their own oxides (e.g., MgO), which may not be as protective. The tendency for oxide formation can be described by the Pilling-Bedworth ratio (PBR), which compares the volume of the oxide to the volume of the metal atoms. For aluminum, Al2O3 has a PBR of approximately 1.28, indicating a protective layer, but for magnesium, MgO has a PBR of 0.81, which is non-protective and prone to cracking. This differential behavior complicates the oxidation dynamics in aluminum alloys used for shell castings.
The oxidation kinetics can be modeled using equations such as the parabolic rate law for diffusion-controlled growth:
$$ \frac{dx}{dt} = \frac{k_p}{x} $$
where \( x \) is the oxide thickness, \( t \) is time, and \( k_p \) is the parabolic rate constant. Integrating this gives:
$$ x^2 = k_p t + C $$
Here, \( C \) is a constant. For aluminum alloys, \( k_p \) depends on temperature and alloy composition. In shell castings, the melt temperature during pouring typically ranges from 700°C to 750°C, accelerating oxide formation. Additionally, the presence of magnesium can alter \( k_p \) due to the formation of spinels like MgAl2O4, which have different growth rates. The total oxide content in the melt can be estimated from factors like holding time and surface area exposure, contributing to inclusion formation in shell castings.
During mold filling, the fluid dynamics play a crucial role in defect generation. For thin-walled shell castings with complex blade arrays, the flow resistance is high, often described by the Reynolds number (Re) to characterize flow regime:
$$ Re = \frac{\rho v D}{\mu} $$
where \( \rho \) is the melt density (approximately 2400 kg/m³ for aluminum alloys), \( v \) is the flow velocity, \( D \) is the hydraulic diameter, and \( \mu \) is the dynamic viscosity (around 0.0013 Pa·s at pouring temperatures). To ensure complete filling, velocities may be increased, but if Re exceeds 2000, turbulent flow can occur, leading to oxide entrainment. The critical velocity for avoiding turbulence can be derived from Bernoulli’s principle and viscosity effects, but in practice, for shell castings, the narrow passages exacerbate turbulence. This results in the folding of oxide films into the melt, which then become trapped at locations like blade roots, where sectional changes cause flow separation and inclusion deposition. The probability of inclusion entrapment \( P_i \) can be expressed as a function of flow parameters:
$$ P_i = f(Re, \text{geometry}, \text{oxide concentration}) $$
where geometry refers to the complex shape of shell castings. Computational fluid dynamics (CFD) simulations often reveal that regions with high shear rates, such as blade roots, are prone to inclusion accumulation, aligning with the observed defect locations in shell castings.
The composition of the oxide inclusions varies based on the alloy system. For typical aluminum-silicon-magnesium alloys used in shell castings, the inclusions may comprise Al2O3, MgO, SiO2, and complex spinels. The EDS data from the inclusions show high oxygen and aluminum, with significant magnesium, suggesting a mix of oxides. The formation of these phases can be thermodynamically predicted using Ellingham diagrams or activity coefficients. For instance, the Gibbs free energy change for alumina formation is:
$$ \Delta G_{\text{Al}_2\text{O}_3} = -RT \ln K_{\text{eq}} $$
where \( R \) is the gas constant, \( T \) is temperature, and \( K_{\text{eq}} \) is the equilibrium constant. At casting temperatures, \( \Delta G \) is highly negative, favoring oxidation. Similarly, for magnesium oxide:
$$ 2\text{Mg} + \text{O}_2 \rightarrow 2\text{MgO}, \quad \Delta G < 0 $$
The interplay between these reactions depends on the melt composition, which in shell castings is carefully controlled but can vary due to recycling or impurity introduction. The table below summarizes common oxide phases and their properties in aluminum alloy shell castings:
| Oxide Phase | Chemical Formula | Density (g/cm³) | Melting Point (°C) | Pilling-Bedworth Ratio | Typical Source in Shell Castings |
|---|---|---|---|---|---|
| Alumina | Al2O3 | 3.95 | 2072 | 1.28 | Surface oxidation, impurity |
| Magnesia | MgO | 3.58 | 2852 | 0.81 | Mg addition, recycling |
| Silica | SiO2 | 2.65 | 1713 | 2.15 | Sand erosion, alloying |
| Spinel | MgAl2O4 | 3.58 | 2135 | ~1.0 | Reaction of Al2O3 and MgO |
These oxides, once incorporated into the melt, act as stress concentrators. During machining of shell castings, external forces cause the brittle oxides to deform and create linear indications on the surface. The mechanism can be modeled using fracture mechanics: the stress intensity factor \( K \) at an inclusion tip is given by:
$$ K = Y \sigma \sqrt{\pi a} $$
where \( Y \) is a geometric factor, \( \sigma \) is the applied stress, and \( a \) is the inclusion size. If \( K \) exceeds the fracture toughness of the oxide, it may crack, but in this case, the oxide remains intact but becomes visible due to plastic deformation of the surrounding aluminum matrix. This explains why fluorescent inspection, which relies on surface discontinuities, detects these defects post-machining in shell castings.
Preventing these linear defects requires a multi-faceted approach targeting the melting, pouring, and solidification stages of shell castings production. First, raw material purity is essential. Using high-purity primary aluminum and master alloys minimizes initial oxide content. Recycled material, while economical, should be limited due to its higher inclusion load. All charge materials must be clean and dry to avoid introducing moisture, which decomposes to form oxides. Second, melting practices should be optimized. Stirring should be gentle, using tools designed to minimize surface disruption, and performed under protective atmospheres like argon or nitrogen to reduce oxidation. Fluxing and degassing are critical; efficient rotary degassing can remove dissolved hydrogen and float oxides to the slag. The efficiency of degassing can be quantified by the rate of hydrogen removal:
$$ \frac{dC}{dt} = -k (C – C_{\text{eq}}) $$
where \( C \) is the hydrogen concentration, \( k \) is the mass transfer coefficient, and \( C_{\text{eq}} \) is the equilibrium concentration. Proper degassing reduces porosity and inclusion levels in shell castings.
Third, pouring system design must ensure laminar flow. For shell castings with thin sections, gating ratios should be calculated to control velocity. The use of ceramic filters in the runner system can trap inclusions before the metal enters the mold cavity. The filtration efficiency \( \eta \) for a porous filter is given by:
$$ \eta = 1 – \exp\left(-\frac{\alpha L}{d_p}\right) $$
where \( \alpha \) is a capture coefficient, \( L \) is filter thickness, and \( d_p \) is particle size. Implementing such filters significantly improves the cleanliness of shell castings. Additionally, mold design can incorporate vents to reduce backpressure and promote steady filling. Simulation tools like MAGMA or ProCAST can predict flow patterns and optimize these parameters for shell castings.
Fourth, process monitoring is key. Real-time methods such as LiMCA (Liquid Metal Cleanliness Analyzer) can assess inclusion counts in the melt, allowing for corrective actions before pouring. Statistical process control charts can track variables like melt temperature, holding time, and pouring speed, ensuring consistency in shell castings production. The table below outlines key preventive measures and their impact on defect reduction in shell castings:
| Preventive Measure | Description | Targeted Defect Cause | Expected Reduction in Linear Defects |
|---|---|---|---|
| High-purity charge materials | Use of primary Al and clean master alloys | Reduces initial oxide sources | 20-30% |
| Controlled stirring | Gentle stirring under inert atmosphere | Minimizes oxide entrainment | 15-25% |
| Advanced degassing | Rotary degassing with argon | Removes dissolved gases and oxides | 25-35% |
| Optimized gating | Design for laminar flow, use of filters | Prevents turbulence and inclusion entrapment | 30-40% |
| Process monitoring | Real-time inclusion detection and SPC | Ensures consistency and early intervention | 10-20% |
Implementing these measures holistically can drastically reduce the incidence of linear defects, enhancing the yield and performance of aluminum alloy shell castings. It is also worth noting that post-casting treatments, such as heat treatment, can affect defect visibility. For instance, solution heat treatment may diffuse elements but not eliminate large oxides. Therefore, prevention at the casting stage is paramount for shell castings.
In conclusion, the linear defects observed in aluminum alloy shell castings are not cracks but manifestations of internal oxide inclusions deformed during machining. Through detailed SEM and EDS analysis, I have characterized these defects as wrinkled oxide films with high oxygen and magnesium content, originating from melting and pouring processes. The complex geometry of shell castings exacerbates turbulence and inclusion entrapment, particularly at blade roots. By applying principles from fluid dynamics, oxidation kinetics, and fracture mechanics, I have explained the defect formation mechanism. To mitigate these issues, a combination of material purity, process control, and design optimization is essential. As the demand for high-integrity shell castings grows in aerospace and other industries, continued research into advanced casting techniques and real-time monitoring will further improve quality. This analysis underscores the importance of a thorough understanding of metallurgical processes in producing reliable aluminum alloy shell castings, ensuring they meet the stringent standards required for critical applications.
Future work could explore non-destructive evaluation methods, such as micro-computed tomography, to detect inclusions before machining in shell castings. Additionally, alloy modifications to enhance oxide stability or the development of novel refining agents may offer new avenues for defect prevention. Ultimately, the goal is to achieve near-perfect shell castings with minimal defects, leveraging insights from this study to drive innovation in aluminum casting technology.
