In my research, I focused on addressing the environmental challenges posed by Bayer process red mud, a solid waste generated during alumina production. Red mud contains significant amounts of strong alkaline substances and is classified as hazardous material, with annual emissions creating immense pressure on ecosystems due to land occupation, alkali leakage, and air pollution from fine particles. To mitigate these issues, I explored a sustainable approach to utilize red mud by extracting valuable metals, particularly iron, through carbothermic reduction. The goal was to produce high-performance abrasion-resistant white cast iron, which can be used in applications like wire drawing dies and grinding balls. This study involved pre-treating red mud via disc pelletization to improve its handling during smelting, followed by molten-state carbothermic reduction in an induction furnace. I then analyzed the microstructure, hardness, impact toughness, and wear resistance of the resulting white cast iron. The findings demonstrate that this method effectively transforms red mud into a useful material, contributing to waste reduction and resource recovery.
The red mud used in this experiment was sourced from an alumina plant in China, with its fine particle size distribution posing challenges for direct smelting. As shown in Table 1, the chemical composition includes iron oxide (Fe₂O₃) at 23.95% by weight, along with alumina (Al₂O₃), calcium oxide (CaO), silica (SiO₂), and other oxides. The particle size analysis revealed that over 82% of particles are below 10 μm, making pelletization essential to prevent operational issues like poor permeability and furnace explosions. For reduction, I used coke as a carbon source, with its composition detailed in Table 2. The disc pelletization process was employed to agglomerate the red mud and coke mixture, enhancing the smelting efficiency. This pre-treatment involved mixing the powders, adding water as a binder, and rolling them in a disc pelletizer to form cold-solid pellets. The pellets were then dried and subjected to molten-state carbothermic reduction in an induction furnace, where temperatures reached 1,600°C to facilitate the reduction reactions. After reduction, the molten white cast iron was cast into samples for further analysis, while the slag was separated and examined.
| Component | Content |
|---|---|
| Fe₂O₃ | 34.21 |
| Al₂O₃ | 19.10 |
| CaO | 18.85 |
| SiO₂ | 12.20 |
| Na₂O | 5.00 |
| TiO₂ | 5.10 |
| Cr₂O₃ | 0.22 |
| MnO | 0.1 |
| NiO | 0.01 |
| MgO | 0.5 |
| Component | Content |
|---|---|
| Fixed Carbon | 83.0 |
| Ash | 11.31 |
| Volatile Matter | 3.3 |
| Sulfur | 0.71 |
| Moisture | 1.05 |
The disc pelletization process proved effective, with pellets achieving a yield of 61.3% for sizes between 5 mm and 16 mm, as summarized in Table 3. The green pellet strength was measured at 13.43 N per pellet, and the drop strength reached 24.25 times per pellet, both exceeding industrial requirements. This success is attributed to the fine particle size of red mud, which increases surface area and enhances bonding through capillary forces. During smelting, the pellets exhibited good thermal stability, maintaining porosity until melting, which ensured smooth reduction. The carbothermic reduction reactions involved the stepwise reduction of iron oxides and other metal oxides. Based on thermodynamic principles, the Gibbs free energy changes for key reactions were calculated to determine their spontaneity. For instance, the reduction of iron oxides proceeds as follows:
$$3\text{Fe}_2\text{O}_3(s) + \text{C}(s) = 2\text{Fe}_3\text{O}_4(s) + \text{CO}(g), \quad \Delta_r G_m^\theta = 1.1672 – 257.76T \, \text{J·mol}^{-1}$$
$$\text{Fe}_3\text{O}_4(s) + \text{C}(s) = 3\text{FeO}(s) + \text{CO}(g), \quad \Delta_r G_m^\theta = 206343 – 215.37T \, \text{J·mol}^{-1}$$
$$\text{FeO}(s) + \text{C}(s) = \text{Fe}(s) + \text{CO}(g), \quad \Delta_r G_m^\theta = 157827 – 157.93T \, \text{J·mol}^{-1}$$
Additionally, oxides of nickel, manganese, and chromium were reduced, contributing to the alloying elements in the white cast iron. The relationship between Gibbs free energy and temperature, illustrated in Figure 5 of the original study, indicates that these reactions become spontaneous above certain temperatures, such as 704.81 K for Fe₂O₃ reduction. The final products included a white cast iron and a slag, with their compositions analyzed in Tables 5 and 6. The white cast iron was found to be hypoeutectic, with a carbon content of 3.68 wt% and alloying elements like chromium (0.423 wt%), resulting in a microstructure devoid of graphite. The slag had a basicity ratio (CaO+MgO)/SiO₂ of 1.35 and low residual iron content (1.40 wt%), confirming efficient reduction.
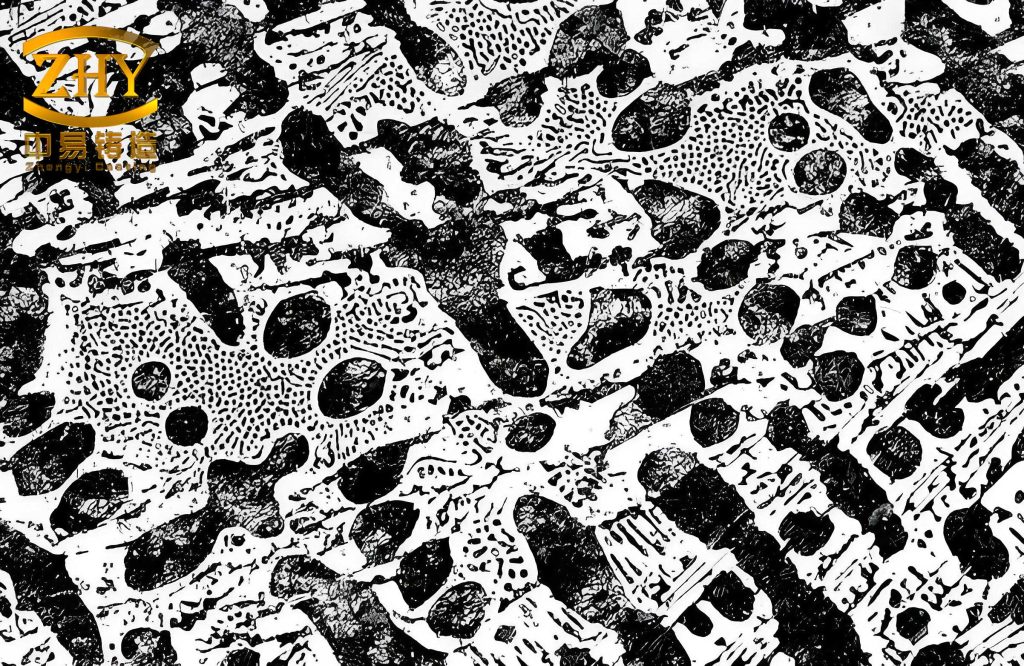
The microstructure of the white cast iron was examined using optical and scanning electron microscopy. It revealed a hypoeutectic structure consisting of pearlite and ledeburite, with no coarse primary cementite network or graphite formation. The white cast iron exhibited a bright white fracture surface, typical of such materials. In high-magnification images, the pearlite appeared as a lamellar mixture of ferrite and cementite, formed from austenite decomposition during cooling, while the ledeburite comprised fine pearlite dispersed in a cementite matrix. This microstructure is characteristic of abrasion-resistant white cast iron, which derives its properties from hard phases like cementite. The hardness of the white cast iron was measured using a Rockwell hardness tester, yielding an average value of HRC 57.58, as shown in Table 7. This high hardness is advantageous for wear-resistant applications, but it often correlates with brittleness. To assess this, I conducted impact toughness tests on unnotched specimens, obtaining an impact toughness of 5.239 J/cm². The load-time curve from the impact test indicated a rapid crack propagation after initiation, suggesting brittle fracture behavior, which is common in high-hardness white cast iron.
| Parameter | Value |
|---|---|
| Pellet Yield (5–16 mm diameter) | 61.3% |
| Green Pellet Compressive Strength | 13.43 N/pellet |
| Green Pellet Drop Strength | 24.25 times/pellet |
| Element | Content |
|---|---|
| C | 3.68 |
| Si | 0.375 |
| Ni | 0.092 |
| Cr | 0.423 |
| Mn | 0.221 |
| Ti | 0.145 |
| S | 0.078 |
| P | 0.085 |
| Fe | Balance |
| Component | Content |
|---|---|
| TFe | 1.40 |
| SiO₂ | 21.53 |
| Al₂O₃ | 32.81 |
| CaO | 28.17 |
| MgO | 0.94 |
| Na₂O | 5.25 |
Wear resistance is a critical property for white cast iron, especially in mining and industrial applications. I evaluated the wear performance using a abrasive wear tester under controlled conditions. The results, summarized in Table 8, showed a wear rate of 0.563%, indicating excellent wear resistance. This can be attributed to the hard cementite phase in the microstructure, which protrudes during wear as the softer pearlite matrix erodes, thereby protecting the underlying material. The combination of high hardness and good wear resistance makes this white cast iron suitable for demanding environments. However, the relatively low impact toughness highlights a trade-off, which may limit its use in applications requiring high shock loads. To optimize the white cast iron properties, further studies could explore adjustments in composition or heat treatment. For instance, varying the carbon content or adding alloying elements like molybdenum might enhance toughness without compromising hardness. Additionally, the slag from the reduction process could be investigated for secondary uses, such as in construction materials, to achieve a zero-waste approach.
| Property | Value |
|---|---|
| Hardness (HRC) | 57.58 |
| Impact Toughness (aK) | 5.239 J/cm² |
| Parameter | Value |
|---|---|
| Initial Mass | 2.665 g |
| Final Mass | 2.650 g |
| Wear Loss | 0.015 g |
| Wear Rate | 0.563% |
The economic and environmental implications of this process are significant. By converting red mud into white cast iron, we not only reduce waste disposal costs but also produce a valuable material that can replace conventional abrasion-resistant alloys. The disc pelletization step is cost-effective and scalable, aligning with industrial practices for iron ore processing. Moreover, the use of an induction furnace allows for precise temperature control and efficient energy utilization. In terms of sustainability, this method contributes to circular economy principles by recovering iron from a hazardous by-product. Future work should focus on pilot-scale trials to assess technical feasibility and economic viability. Potential challenges include variability in red mud composition and the need for slag management. Nevertheless, this study demonstrates a promising pathway for red mud utilization, with white cast iron as a key output. The successful production of high-hardness white cast iron underscores the potential of this approach in metallurgical applications.
In conclusion, my research shows that Bayer process red mud can be effectively processed through disc pelletization and molten-state carbothermic reduction to produce abrasion-resistant white cast iron. The white cast iron obtained exhibits a hypoeutectic microstructure with high hardness (HRC 57.58), reasonable impact toughness (5.239 J/cm²), and excellent wear resistance. This white cast iron is suitable for use in wear-prone components, offering an eco-friendly solution to red mud accumulation. The process also yields a slag with low iron content, indicating efficient reduction. Overall, this work highlights the feasibility of transforming industrial waste into high-performance materials, with white cast iron serving as a prime example of value recovery. Further optimization could enhance the properties of white cast iron, making it even more competitive in the market. As global emphasis on sustainability grows, such innovative approaches will play a crucial role in resource conservation and environmental protection.
To delve deeper into the thermodynamic aspects, I derived the Gibbs free energy equations for the reduction reactions. For the reduction of chromium oxide, which contributes to the alloying in white cast iron, the reaction is:
$$\text{Cr}_2\text{O}_3(s) + 3\text{C}(s) = 2\text{Cr}(s) + 3\text{CO}(g), \quad \Delta_r G_m^\theta = 784377 – 522.29T \, \text{J·mol}^{-1}$$
The temperature at which this reaction becomes spontaneous can be calculated by setting $\Delta_r G_m^\theta = 0$, yielding $T \approx 1501.8$ K. Similarly, for manganese oxide reduction, which also influences the white cast iron properties:
$$\text{MnO}(s) + \text{C}(s) = \text{Mn}(s) + \text{CO}(g), \quad \Delta_r G_m^\theta = 270690 – 159.52T \, \text{J·mol}^{-1}$$
This reaction starts at about 1696.9 K. These calculations help in understanding the reduction sequence during smelting. In practice, the high temperature of 1,600°C (1,873 K) in the induction furnace ensures that all these reactions proceed efficiently, leading to a well-alloyed white cast iron. The presence of chromium and manganese in the white cast iron enhances its hardenability and wear resistance, which are desirable traits for abrasion-resistant applications. Additionally, the carbon content in the white cast iron is critical for forming cementite, the hard phase that imparts wear resistance. The balance between carbon and alloying elements determines the final microstructure and properties of the white cast iron.
From a microstructural perspective, the white cast iron produced is classified as hypoeutectic because its carbon content (3.68 wt%) is below the eutectic point of approximately 4.3 wt% for the Fe-C system. This results in the formation of primary austenite upon solidification, which later transforms to pearlite, and a eutectic mixture of ledeburite. The absence of graphite is due to the rapid cooling and the presence of carbide-stabilizing elements like chromium. This microstructure is ideal for wear resistance, as cementite provides a hard barrier against abrasion. However, the brittleness associated with cementite can be mitigated by controlling the cooling rate or through alloy design. For instance, adding nickel or copper might improve toughness while maintaining the hardness of the white cast iron. In my experiments, the white cast iron showed a good combination of properties, but there is room for improvement through further alloying or heat treatment processes.
The wear mechanism in white cast iron involves a complex interaction between the hard cementite phase and the softer matrix. During abrasive wear, the pearlite matrix wears away first, causing the cementite to protrude and bear the load. This reduces further wear and extends the service life of components made from this white cast iron. The wear rate of 0.563% observed in my tests is comparable to that of commercial abrasion-resistant white cast irons, validating the effectiveness of this production method. To put this into perspective, traditional white cast iron used in mining equipment often has wear rates in the range of 0.5–1.0%, depending on the application. Thus, the white cast iron from red mud reduction is competitive in performance. Moreover, the environmental benefits of using red mud as a raw material add value beyond technical specifications. This aligns with global trends towards green manufacturing and sustainable resource use.
In summary, the production of white cast iron from Bayer process red mud is a technically viable and environmentally sound process. The key steps—pelletization, carbothermic reduction, and casting—yield a high-quality white cast iron with desirable properties for abrasion-resistant applications. The white cast iron’s microstructure, dominated by pearlite and ledeburite, ensures high hardness and wear resistance, while the impact toughness is acceptable for many industrial uses. Future research could explore the effects of varying red mud sources or incorporating post-treatment processes like annealing to enhance toughness. Additionally, life cycle assessments could quantify the environmental savings compared to conventional white cast iron production. Overall, this work contributes to the broader goal of waste valorization, with white cast iron standing out as a promising product. By continuing to refine this process, we can turn a hazardous waste into a valuable resource, promoting a circular economy in the metallurgical industry.
