Figure (a) shows the effect of natural aging on the hardness of high chromium cast iron. It can be seen from figure (a) that at 950 ℃, after room temperature aging treatment, the macro hardness of high chromium cast iron increases from 63.6 HRC to 67 HRC, while the micro hardness decreases from 881.2 hv to 736.6 HV. The same experimental phenomenon was observed at 900 ℃ and 1000 ℃. At 900 ℃, the macro hardness increased from 62.9 HRC to 66 HRC, and the micro hardness decreased from 826.9 hv to 743.8 HV; At 1000 ℃, the macro hardness increased from 61.8 HRC to 64.5 HRC, and the micro hardness decreased from 801 hv to 713.8 HV.
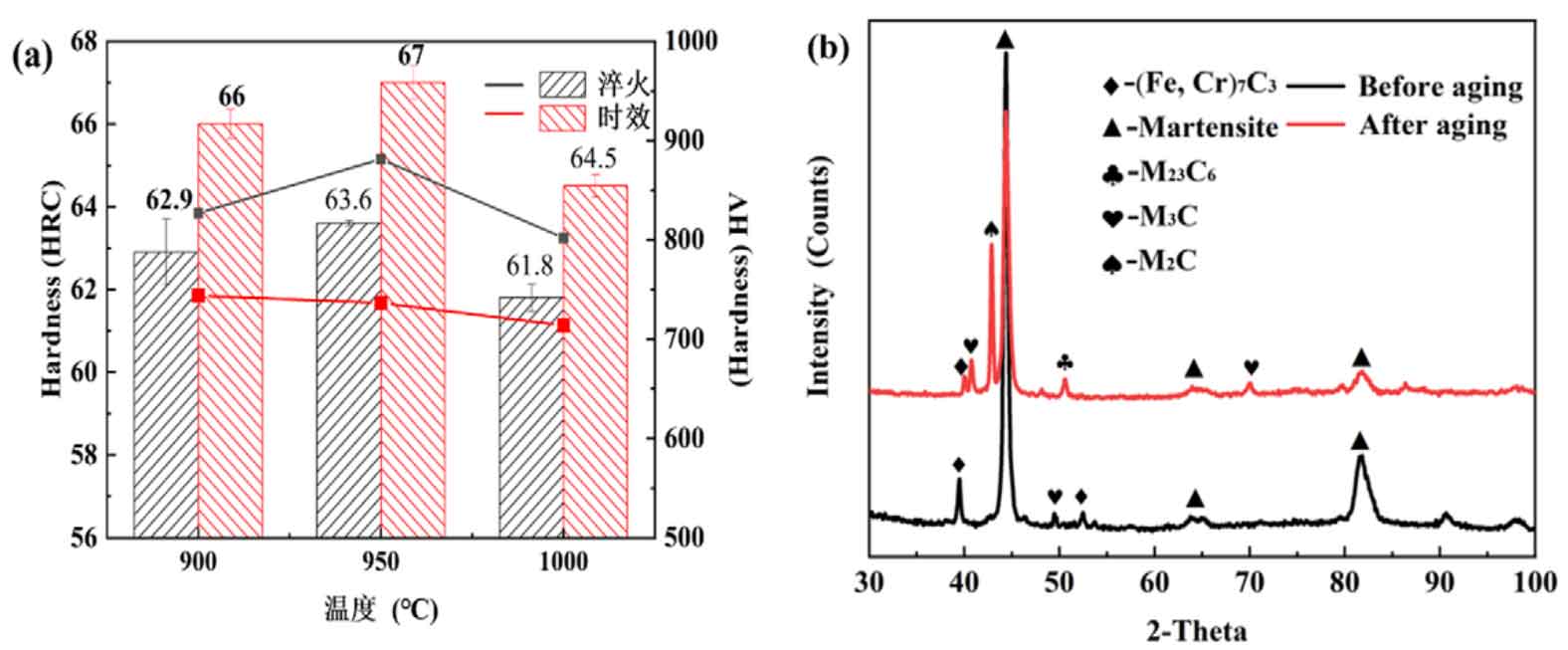
(b) spectra of the investigated at 950 ℃
Figure (b) shows the XRD scanning phase analysis results. When quenched at 950 ℃, the XRD diffraction peaks mainly include martensite peak, M7C3 carbide peak, M23C6 carbide peak and m3c carbide peak. After natural aging, there are M2C carbide peaks in addition to martensite peak, M7C3 carbide peak, M23C6 peak and m3c peak. That is, comparing the XRD phase composition of 950 ℃ quenching and natural aging, it can be found that M2C carbide precipitates after natural aging treatment of high chromium cast iron.
After quenching at 900 ℃ – 1000 ℃, the matrix structure of high chromium cast iron is martensite. Relevant studies show that precipitation occurs in martensite during natural aging ε- M2C or η- M2C carbide. because η- M2C carbide ratio ε- M2C carbide is stable, ε- M2C can be further transformed into η- M2C carbide. The reason is that, contrary to the “solidification” hypothesis, carbon atoms can also move and diffuse at room temperature. When the carbon content in martensite of high chromium cast iron exceeds 0.6 wt.%, martensite is hexagonal structure, which is unstable. However, the newly formed untempered hexagonal martensite after quenching will decompose into cubic structure during room temperature aging. In this process, the carbon atoms in the martensite of high chromium cast iron will redistribute and gather at the defects, so as to form a carbon rich / carbon poor amplitude modulation structure. During further relaxation, the enriched carbon atoms are removed from the matrix. When the chemical composition of the carbon rich region reaches M2C, the carbon atoms will rearrange spontaneously ε- Or η- Therefore, M2C carbide precipitates through further relaxation. In this experiment, the interface between the matrix and the eutectic carbide M7C3 becomes a defect structure due to the large difference in the chemical composition of C and Cr. In the process of unstable decomposition, the carbon atoms in the martensite of high chromium cast iron first enrich to the junction. When the carbon atoms at the junction reach the M2C composition, M2C carbides precipitate at the junction.
Instability decomposition will produce strong elastic interaction in high-carbon and low-carbon regions. Secondary carbides precipitate during quenching and cooling, and stress will also occur in this process. High chromium cast iron MF = 54.8 ℃, higher than room temperature. All these may be conducive to the precipitation of M2C carbides in martensite at room temperature. After natural aging treatment, M2C carbide precipitates from martensite, which leads to precipitation strengthening and increases the macro hardness of high chromium cast iron; However, carbide precipitation will reduce the carbon content in martensitic matrix, which will reduce the microhardness of martensitic matrix. The increase of hardness caused by precipitation strengthening is greater than the decrease of microhardness, so the macro hardness of high chromium cast iron increases after natural aging.
