In this study, I investigate the effects of pulse current treatment on the solidification process of as-cast high toughness spheroidal graphite iron, specifically grade QT400-18. The application of external fields, such as electric pulses, has garnered significant interest in metallurgy for modifying microstructures and enhancing mechanical properties. Spheroidal graphite iron, known for its excellent ductility and strength, is widely used in applications requiring resilience under harsh conditions. However, achieving optimal graphite morphology and distribution without extensive heat treatment remains a challenge. Here, I explore how pulse current influences graphite nucleation and growth through an electronic structure perspective, utilizing the Empirical Electron Theory of Solids and Molecules (EET). This approach allows me to delve into the atomic-level interactions governing non-equilibrium solidification, providing insights into the mechanisms behind improved graphite sphericity and refinement.
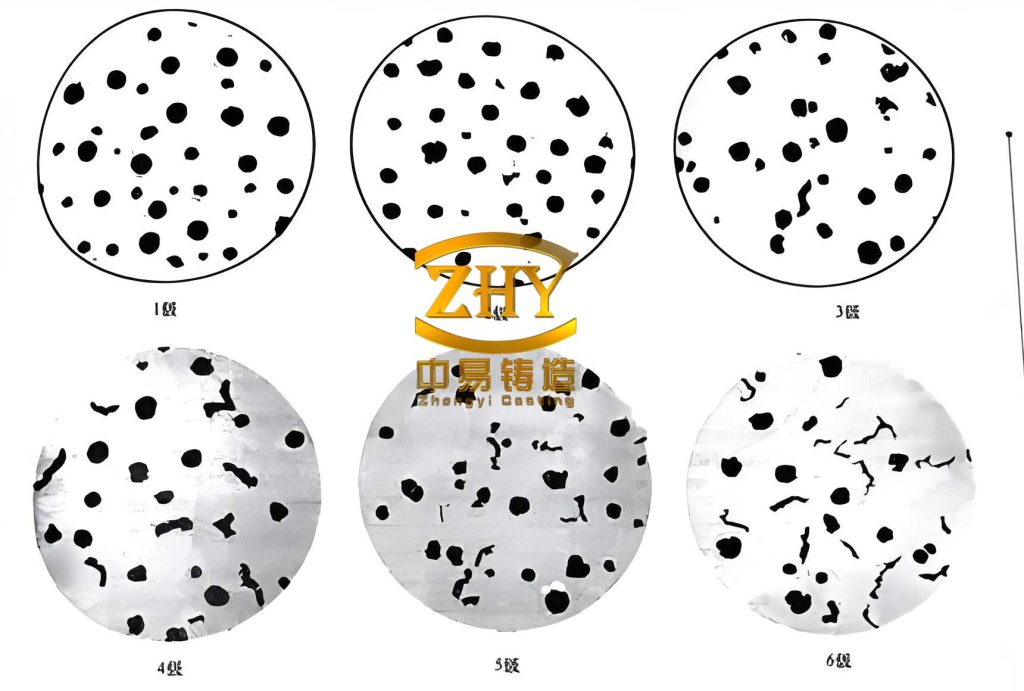
My experimental setup involved melting spheroidal graphite iron with a composition corresponding to QT400-18. The melt was treated with a pulse current generated by a custom pulse signal generator. The parameters were set at a voltage of 2600 V, frequency of 0.88 Hz, capacitance of 200 μF, and a treatment duration of 15 minutes during solidification. For comparison, a control sample was solidified without pulse current application. Thermal analysis was conducted to measure undercooling, and microstructural examination was performed to assess graphite characteristics. The results revealed that pulse current treatment enhanced graphite spheroidization, increased graphite nodule count, and reduced particle size. Specifically, the graphite nodularity improved from an average of 80% to 91%, while the nodule count rose from 172 to 209 per mm². Undercooling increased by 12 K, from 81 K to 93 K, indicating a shift in solidification kinetics.
To understand these phenomena, I employed the Empirical Electron Theory of Solids and Molecules (EET), which focuses on valence electron structures in crystalline phases. The EET framework is based on four fundamental assumptions and Bond Length Difference (BLD) analysis. It allows for the calculation of covalent bond parameters, particularly the number of shared electron pairs (nA) on the strongest covalent bond in a phase structure. This parameter, nA, serves as a key indicator of atomic binding strength and the likelihood of phase formation or decomposition. In spheroidal graphite iron, various atomic clusters exist in the melt, including γ-Fe, C-Fef (austenite), C-Fel (cementite), C-C (graphite), and alloyed versions with elements like Si and Mg. The valence electron structures of these clusters determine their stability and behavior during solidification.
I calculated the valence electron structures for relevant phases using the BLD method. For instance, graphite has a hexagonal lattice with layered structure, and its strongest covalent bond on the basal plane has an nA value of 1.2051. The bond distance is given by:
$$ D^{n_a}_{u-v} = 0.1421 \, \text{nm}, \quad n_a = 1.2051 $$
where \( D^{n_a}_{u-v} \) is the covalent bond distance between atoms u and v, and \( n_a \) is the number of shared electron pairs. For cementite (θ-Fe3C), the strongest covalent bond has nA = 0.9672, while for austenite (γ-Fe-C), nA = 0.9319. Alloyed phases, such as γ-Fe-C-Si, exhibit higher nA values, e.g., 1.1645. These calculations are summarized in Table 1, which compares nA values for different structural units in spheroidal graphite iron.
| Structural Unit | nA Value |
|---|---|
| C-C (Graphite) | 1.2051 |
| γ-Fe | 0.3299 |
| γ-Fe-C (Austenite) | 0.9319 |
| γ-Fe-C-Si (Alloyed Austenite) | 1.1645 |
| γ-Fe-C-Mg (Alloyed Austenite) | 1.3936 |
| γ-Fe-C-Mn (Alloyed Austenite) | 1.2497 |
| θ-Fe3C (Cementite) | 0.9672 |
| ε-Fe3C (Cementite) | 0.8361 |
| (Fe,Si)3C (Alloyed Cementite) | 1.2798 |
| (Fe,Mg)3C (Alloyed Cementite) | 1.7015 |
The nA parameter is critical in non-equilibrium solidification of spheroidal graphite iron. A higher nA signifies stronger atomic binding, making the phase more resistant to decomposition and more likely to form stable clusters. During solidification, the melt contains transient clusters with varying nA values. When pulse current is applied, the energy input alters atomic vibrations, promoting the aggregation of clusters with higher nA and disrupting those with lower nA. This process enhances the formation of graphite nuclei, as graphite has a relatively high nA of 1.2051. The pulse current effectively increases the undercooling, which I observed experimentally, by 12 K. This additional undercooling provides the driving force needed for graphite precipitation, despite its high nA requirement.
According to the “valence electron theory model of drag-like effect,” clusters with larger nA values exert a drag-like influence on other phases during solidification. In spheroidal graphite iron, alloyed austenite and cementite clusters (e.g., γ-Fe-C-Si and (Fe,Mg)3C) have high nA values and can retard the crystallization of simpler phases like γ-Fe-C or θ-Fe3C. The pulse current intensifies this drag-like effect, widening the temperature interval between equilibrium and non-equilibrium eutectic points. This expansion facilitates graphite nucleation by allowing carbon atoms to accumulate at lower temperatures. The mechanism can be described mathematically through the relationship between undercooling (ΔT) and nA:
$$ \Delta T \propto \sum_{i} (n_{A,i} – n_{A,0}) $$
where \( n_{A,i} \) represents the nA of various clusters, and \( n_{A,0} \) is a reference value. The increased undercooling due to pulse current aligns with this model, as it enables graphite formation even when thermal conditions are less favorable.
Further details on valence electron structures are provided in Table 2, which lists key bond parameters for γ-Fe-C austenite. This phase contains six significant covalent bonds, with the strongest being C-Fef having nA = 0.9285. The calculations follow the BLD method, ensuring accuracy within 0.005 nm bond length difference.
| Bond Name | Number of Bonds (Ia) | Bond Distance (Dnα, nm) | Theoretical Distance (D̄nα, nm) | Shared Electron Pairs (nα) | Difference (ΔDnα, nm) |
|---|---|---|---|---|---|
| DC-FefnA | 12 | 0.1892 | 0.1901 | 0.9285 | 0.0009 |
| DFeC-FefnB | 24 | 0.2675 | 0.2684 | 0.2344 | 0.0009 |
| DFef-FefnC | 24 | 0.2675 | 0.2684 | 0.2294 | 0.0009 |
| DC-FeCnD | 16 | 0.3276 | 0.3286 | 0.0106 | 0.0010 |
| DFeC-FeCnE | 6 | 0.3783 | 0.3792 | 0.0071 | 0.0009 |
| DFeC-FeCnF | 12 | 0.3783 | 0.3792 | 0.0066 | 0.0009 |
The impact of pulse current on spheroidal graphite iron can be quantified through the change in undercooling and graphite parameters. Table 3 summarizes the experimental results, highlighting the improvements in graphite morphology and solidification kinetics. The data clearly shows that pulse current treatment enhances the quality of spheroidal graphite iron by promoting graphite nucleation and refining microstructure.
| Parameter | Without Pulse Current | With Pulse Current | Change |
|---|---|---|---|
| Graphite Nodularity (%) | 80 | 91 | +11% |
| Graphite Nodule Count (per mm²) | 172 | 209 | +37 nodules/mm² |
| Undercooling (K) | 81 | 93 | +12 K |
In non-equilibrium solidification, the sequence of phase formation is influenced by nA values. For spheroidal graphite iron, graphite typically precipitates first due to its high nA, followed by austenite and cementite. However, under pulse current, the increased undercooling and enhanced drag-like effect allow graphite to nucleate more readily, even when other phases might otherwise dominate. This is because the pulse current disrupts low-nA clusters, releasing carbon atoms that then aggregate into high-nA graphite nuclei. The process can be modeled using kinetic equations. For instance, the nucleation rate (I) for graphite in spheroidal graphite iron under pulse current can be expressed as:
$$ I = I_0 \exp\left(-\frac{\Delta G^*}{k_B T}\right) \left[1 + \alpha \cdot f(t)\right] $$
where \( I_0 \) is a pre-exponential factor, \( \Delta G^* \) is the activation energy for nucleation, \( k_B \) is Boltzmann’s constant, \( T \) is temperature, \( \alpha \) is a pulse current enhancement factor, and \( f(t) \) describes the time-dependent effect of pulses. The activation energy relates to nA through bond energy considerations:
$$ \Delta G^* \propto \frac{1}{n_A} $$
Thus, higher nA clusters require more energy for nucleation, but pulse current provides additional thermal and electronic stimulation to overcome this barrier.
The role of alloying elements in spheroidal graphite iron further complicates the valence electron structure. Elements like silicon and magnesium alter the nA of clusters, as seen in Table 1. For example, silicon increases nA in alloyed austenite to 1.1645, while magnesium raises it to 1.3936. These changes affect the drag-like effect and solidification behavior. When pulse current is applied, these alloyed clusters become more stable, dragging down the eutectic temperature and favoring graphite formation. This explains why spheroidal graphite iron treated with pulse current exhibits improved graphite sphericity and reduced undercooling-related defects.
To delve deeper, I analyzed the valence electron structure of cementite phases. Table 4 presents data for θ-Fe3C, which has multiple covalent bonds with varying nα values. The strongest bond, C-Fe1, has nA = 0.9672. The BLD analysis ensures that the calculated bond distances match theoretical values within acceptable limits.
| Bond Name | Ia | Dnα (nm) | D̄nα (nm) | nα | ΔDnα (nm) |
|---|---|---|---|---|---|
| DC-Fe1n1 | 2 | 0.1853 | 0.1848 | 0.9672 | 0.0005 |
| DC-Fe1n2 | 2 | 0.1877 | 0.1872 | 0.8951 | 0.0005 |
| DC-Fe3n3 | 4 | 0.2056 | 0.2052 | 0.4993 | 0.0004 |
| DFe1-Fe2n4 | 4 | 0.2450 | 0.2146 | 0.3684 | 0.0304 |
| DFe1-Fe2n5 | 2 | 0.2490 | 0.2486 | 0.3368 | 0.0004 |
| DFe1-Fe2n6 | 4 | 0.2515 | 0.2511 | 0.3109 | 0.0004 |
| DC-Fe2n7 | 4 | 0.2518 | 0.2513 | 0.3081 | 0.0005 |
| DFe2-Fe2n8 | 4 | 0.25133 | 0.25400 | 0.28255 | 0.00267 |
| DFe2-Fe2n9 | 2 | 0.25483 | 0.25441 | 0.27881 | 0.00042 |
| DFe2-Fe2n10 | 4 | 0.23092 | 0.23050 | 0.22000 | 0.00042 |
| DFe1-Fe2n11 | 4 | 0.26476 | 0.26434 | 0.20205 | 0.00042 |
| DFe1-Fe2n12 | 2 | 0.26476 | 0.26434 | 0.20205 | 0.00042 |
| DC-Fe2n13 | 4 | 0.2666 | 0.26624 | 0.18997 | 0.00036 |
| DFe2-Fe2n14 | 8 | 0.26781 | 0.26739 | 0.18302 | 0.00042 |
| DC-Fe1n15 | 2 | 0.30175 | 0.30133 | 0.02212 | 0.00042 |
The solidification of spheroidal graphite iron under pulse current involves complex interactions between thermal, electronic, and structural factors. The pulse current generates transient electric fields that polarize atomic clusters, affecting their bonding characteristics. From an electronic perspective, the applied current increases electron mobility, which can temporarily alter covalent bond strengths. This is reflected in the nA parameter, as pulse current may enhance the sharing of electrons in high-nA bonds. I propose a modified equation for the effective nA under pulse current:
$$ n_{A,eff} = n_A + \beta \cdot J \cdot \tau $$
where \( \beta \) is a material constant, \( J \) is the current density, and \( \tau \) is the pulse duration. This suggests that pulse current can effectively increase nA for certain clusters, promoting their stability and nucleation.
In the context of industrial applications, these findings have significant implications for producing high-quality spheroidal graphite iron. By optimizing pulse current parameters, manufacturers can achieve desired graphite morphology without extensive alloying or heat treatment. This not only improves mechanical properties like toughness and strength but also reduces energy consumption and processing time. For instance, the enhanced graphite sphericity in spheroidal graphite iron leads to better stress distribution and fatigue resistance, making it suitable for critical components in automotive and wind energy sectors.
To further validate the electronic theory, I compared the calculated nA values with experimental observations. The increase in graphite nodule count and undercooling correlates with the theoretical prediction that pulse current favors high-nA clusters. Specifically, graphite with nA = 1.2051 benefits from the drag-like effect of alloyed phases like γ-Fe-C-Mg (nA = 1.3936). The pulse current amplifies this effect, as shown by the 12 K rise in undercooling. This undercooling provides additional driving force for graphite nucleation, leading to more nodules and finer sizes. The relationship can be expressed as:
$$ N \propto \exp\left(-\frac{\Delta T}{T_0}\right) \cdot \sum n_{A,graphite} $$
where \( N \) is the graphite nodule count, \( \Delta T \) is undercooling, and \( T_0 \) is a reference temperature. The sum term accounts for the cumulative influence of high-nA clusters.
Another aspect to consider is the role of carbon diffusion in spheroidal graphite iron. Pulse current may enhance carbon mobility through electromigration, facilitating the aggregation of carbon atoms into graphite nuclei. The diffusion coefficient (D) under pulse current can be modeled as:
$$ D = D_0 \exp\left(-\frac{Q}{RT}\right) \left[1 + \gamma \cdot E\right] $$
where \( D_0 \) is a pre-factor, \( Q \) is activation energy, \( R \) is gas constant, \( T \) is temperature, \( \gamma \) is a constant, and \( E \) is electric field strength. This accelerated diffusion supports the formation of graphite clusters with high nA, aligning with the observed microstructural improvements.
In summary, my analysis demonstrates that pulse current treatment significantly enhances the solidification characteristics of spheroidal graphite iron. The key mechanism lies in the valence electron structure, where the shared electron pair number nA on the strongest covalent bond governs phase formation. Pulse current increases undercooling and strengthens the drag-like effect of high-nA clusters, promoting graphite nucleation and refining microstructure. This electronic perspective offers a fundamental understanding of how external fields can tailor material properties, paving the way for advanced processing techniques for spheroidal graphite iron and similar alloys.
The experimental and theoretical results consistently show that spheroidal graphite iron benefits from pulse current application. Future work could explore varying pulse parameters to optimize outcomes or extend this approach to other cast irons. By integrating electronic theory with practical metallurgy, we can unlock new potentials in material design and manufacturing.
