The figure shows the potentiodynamic polarization curves of three parts of lzqt600-3 ductile iron profile in 3.5% NaCl solution. The corrview software is used to fit the curve by Tafel extrapolation method to obtain the electrode data, so that the corrosion resistance of three parts of the profile in the same corrosion medium can be directly compared by the value. It can be seen from the figure that the shapes of the potentiodynamic polarization curves at the edge, 1 / 2R and the center are similar, indicating that they have similar electrochemical corrosion behaviors. The electrode potential at the edge is more positive than other parts, and the self corrosion potential is the largest.
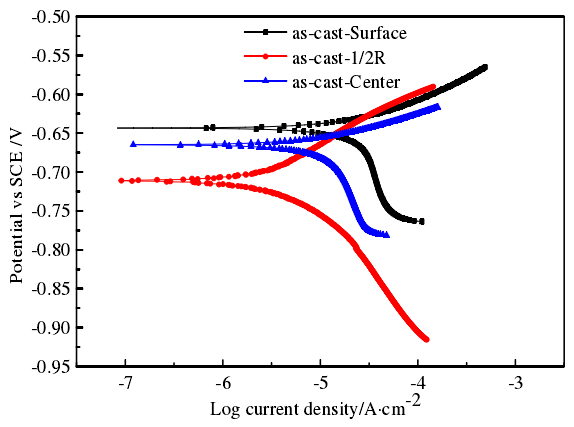
The higher the corrosion potential and the lower the self-corrosion current and self-corrosion rate are, the better the corrosion resistance is. The self corrosion current density of edge, 1 / 2R and core are 6.9743 × 10-5, 0.4069 × 10-5 and 1.6778 × 10-5 amp / cm2, respectively, and the corresponding corrosion rates are 0.0062608, 0.033332 and 0.15062 mm / A, respectively. Therefore, it can be inferred that the corrosion resistance tendency of different parts is: edge > 1 / 2R > center. From the edge to the center, the diameter of graphite balls increases but the number decreases, the number of pearlite increases and the distance between pearlite lamellae increases. 3.5% NaCl solution is easy to enter into the matrix to corrode the sample, so the corrosion resistance of the core is poor.
