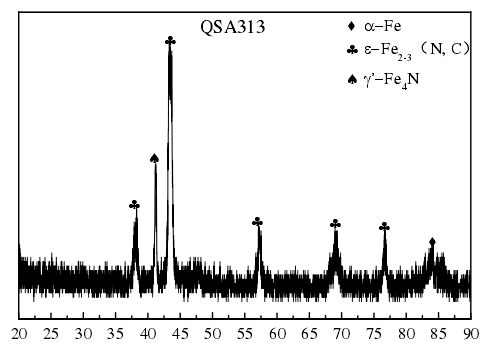
The picture shows the XRD spectrum of the surface of the sample at the edge after being treated at 540 ℃ for 6 h and NH3: CO2 ratio of 4:1. It can be seen from Fig. 4-2 that α – Fe, ε – fe2-3 (n, c) and γ′ – Fe4N phases are formed in the surface layer of the compound. In this process, the content of CO2 is low and the active carbon atom is provided by CO2. The mechanism of nitriding is similar to that of nitriding. There are two reactions in the process of gas nitrocarburizing. One is to decompose the active nitrogen atom at a certain temperature, and the other is to react effectively with Fe matrix. Fe not only promotes the decomposition of NH3, but also adsorbs active nitrogen.
In the initial stage of nitriding, the α – Fe (n) solid solution on the surface of the sample is not saturated, and the layer thickens with the increase of nitriding time. With the increase of active nitrogen atoms in the atmosphere, the N content in the solid solution of α – Fe (n) → γ – Fe (n) → γ′ – Fe4N occurs, and the phase transition of α – Fe (n) → γ′ – Fe4N is formed on the surface. With the increase of nitriding time, the active nitrogen and carbon atoms continuously infiltrate into the matrix, which indicates that after the formation of single γ′ phase, γ′ – Fe4N reaches supersaturation state, and the phase transition of γ′ – Fe4N → ε – fe2-3 (n, c) occurs, and a higher nitrogen content ε phase is formed on the surface of the sample. Therefore, the diffusion layer consists of compound layer composed of ε phase and γ′ phase and diffusion layer composed of α – Fe phase.
However, in theory, the carbon atoms with radius of 0.86 Å are always larger than the lattice gap radius of α – Fe. From the geometric perspective, carbon atoms will not be dissolved in α – Fe, but in reality, with the increase of nitrocarburizing temperature, the solubility of carbon atoms in α – Fe increases gradually from the initial solubility of 0 to 0. Therefore, in the nitrocarburizing temperature of 540 ℃ to 580 ℃, the carbon content on the surface of three parts of lzqt600-3 ductile iron profile is very small. However, the addition of carbon atoms can promote the penetration of nitrogen atoms, which has a positive effect on the increase of the thickness of the layer. However, too much carbon atoms will cause the thickness of the layer to decrease.
